Deliver Assemblon libraries into mammalian genomes at precise locations.
We currently use several different platforms to integrate big DNA into embryonic stem cells, including:
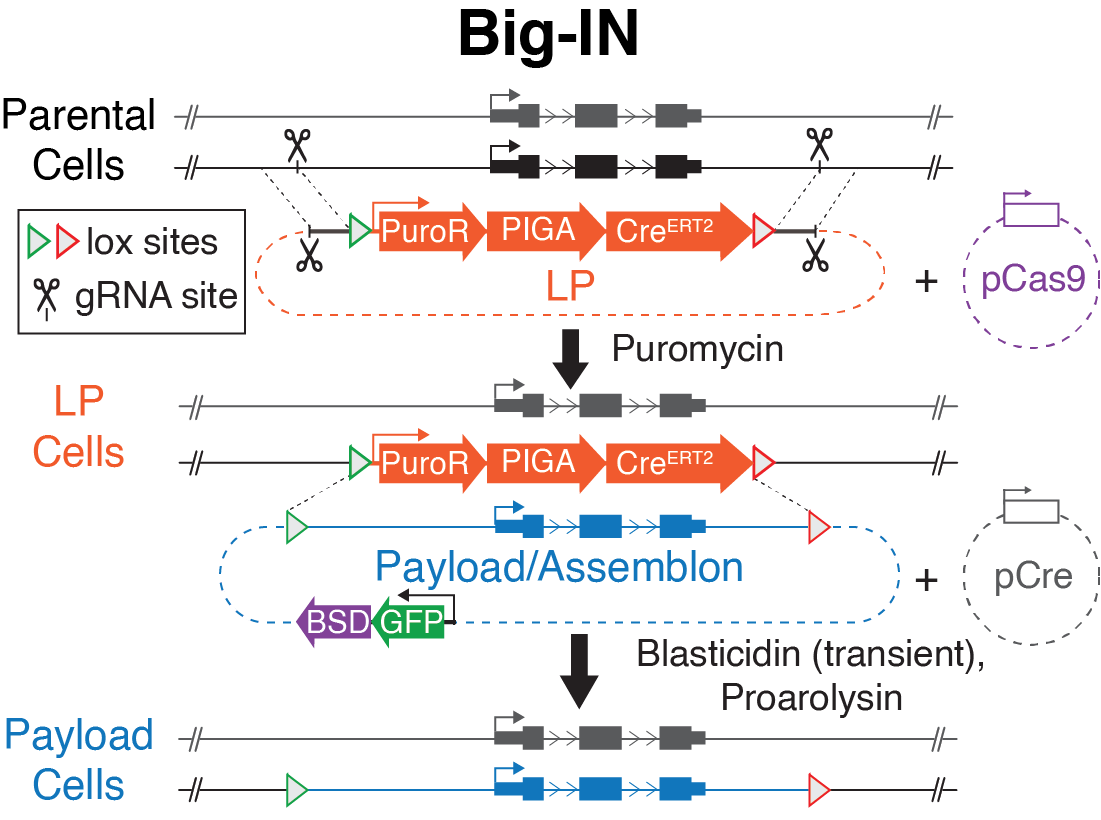
Big-IN (Brosh, Laurent et al., PNAS 2021): For scarless and repeated delivery of big DNAs we developed the “Big-IN” strategy, which enables scarless single-copy integration at defined genomic sites. Briefly, the strategy comprises of 2 steps: Landing Pad integration (mediated by CRISPR-Cas9) followed by Cre-mediated Assemblon recombination (via RMCE, recombinase-mediated cassette exchange). In the first step, one of the alleles of a locus-of-interest is precisely replaced with a Landing Pad containing all the necessary components for enabling site-specific recombination and selection. In the second step, cells are introduced with the synthetic Assemblon, which seamlessly replaces the Landing Pad. Cells in which successful recombination occurred are then isolated thanks to a powerful counterselection strategy that eliminates landing pad harboring parental cells.
We have recently applied Big-IN to rewrite dozens of versions of the mouse Sox2 locus (Brosh et al.. Mol Cell 2023).
mSwAP-In (Zhang et al., Nature 2023): For scarless and iterative genome writing we developed a Big DNA integration system called mammalian Switching Antibiotic markers Progressively for Integration (mSwAP-In). This strategy relies on iterative switching of two selectable marker cassettes (MCs) harboring distinct sets of fluorescence proteins and positive/negative selection markers. To apply mSwAP-In, MC1 is first integrated at the locus of interest (to be rewritten). Then, a synthetic Big DNA payload and MC2 are delivered to cells, and aided by 4 simultaneous CRISPR/Cas9 cuts and homology-directed recombination, they overwrite the endogenous locus. Cells are selected for the gain of MC2 and the loss of MC1. This process can be repeated iteratively and indefinitely to write huge swaths of DNA. A final (and optional) step involved removal of the last MC to render the entire genome writing process scarless.
We have recently applied mSwAP-In to rewrite the mouse Trp53 (p53) gene with a mutation-resistant synthetic version and to generate a mouse model for coronavirus infection (Zhang et al., Nature 2023).
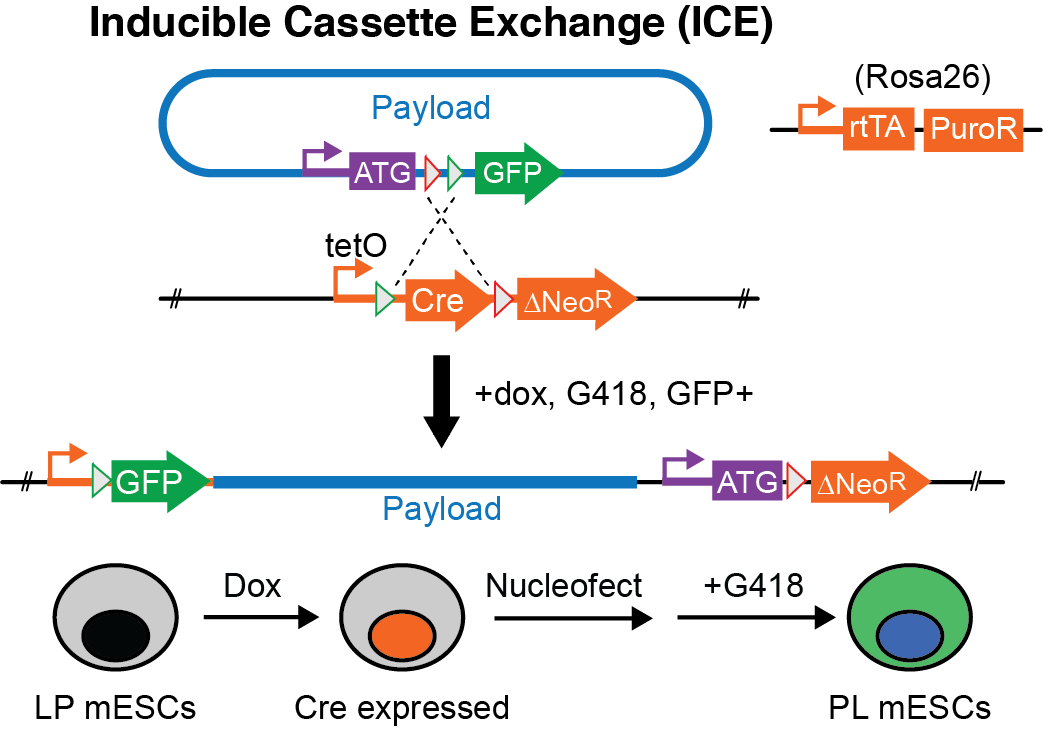
The Inducible Cassette Exchange (ICE) method, first described by Michael Kyba (Iacovino et al., Stem Cells. 2011), is used to integrate big DNA assemblons into mouse ESCs. We have recently successfully implemented this method to integrate a 101 kb HPRT1 gene into mouse embryonic stem cells (Mitchell et al., bioRxiv 2018) as well as to delivery multiple Hoxa variants (Pinglay et al., Science 2022).